|
The abbreviation of the journal title 'Journal of anaesthesiology, clinical pharmacology' is 'J. Anaesthesiol. Clin. Pharmacol.'. It is the recommended abbreviation to be used for abstracting, indexing and referencing purposes and meets all criteria of the ISO 4 standard for abbreviating names of scientific journals.
Abbreviation rules
The table below outlines in detail the ISO 4 rules and matches to the ISSN maintained list of title word abbreviations (TWA) to derived the abbreviation.
About the journalOther journal abbreviations
J Anaesthesiol Clin Pharmacol. 2015 Oct-Dec; 31(4): 450–456.
PMID: 26702198
See commentary 'Ropivacaine: Is it a good choice for spinal anesthesia?' in volume 31 on page 457.
See commentary 'Endotracheal tube leak: What should we do?' in volume 31 on page 459.
This article has been cited by other articles in PMC.
Abstract
Topical anesthetics are being widely used in numerous medical and surgical sub-specialties such as anesthesia, ophthalmology, otorhinolaryngology, dentistry, urology, and aesthetic surgery. They cause superficial loss of pain sensation after direct application. Their delivery and effectiveness can be enhanced by using free bases; by increasing the drug concentration, lowering the melting point; by using physical and chemical permeation enhancers and lipid delivery vesicles. Various topical anesthetic agents available for use are eutectic mixture of local anesthetics, ELA-max, lidocaine, epinephrine, tetracaine, bupivanor, 4% tetracaine, benzocaine, proparacaine, Betacaine-LA, topicaine, lidoderm, S-caine patch™ and local anesthetic peel. While using them, careful attention must be paid to their pharmacology, area and duration of application, age and weight of the patients and possible side-effects.
Keywords: Topical anesthesia, Eutectic mixture of local anesthetics, iontophoresis, local anesthetic, skin permeation enhancer, sonophoresis, uses and side-effects of topical anesthetics
Introduction
Injections of local anesthetics are painful. It can worsen needle anxiety, and can cause tissue edema, which distorts the surgical site. Use of topical anesthesia can avoid all these problems and is becoming a routine in clinical practice.
Topical anesthesia is defined as superficial loss of sensation in conjunctiva, mucous membranes, or skin, produced by direct application of local anesthetic solutions, ointments, gels or sprays.
The first local anesthetic (cocaine) was a topical anesthetic and was serendipitously discovered to have anesthetic properties, when Albert Niemann in 1860, like many chemists of that era tested his newly isolated compound and noted that it caused numbing of the tongue.[] In 1884, Karl Koller, an ophthalmic surgeon, demonstrated that general anesthesia could be avoided for ophthalmic procedures by using cocaine application to the conjunctiva.[] The discovery of various amide and ester local anesthetics, their topical preparations and delivery systems in due course of time opened the gate of immense possible uses of topical anesthetics.
Mechanism of Action
Topical anesthetics reversibly block nerve conduction near their site of administration by targeting free nerve endings in the dermis or mucosa, thereby producing temporary loss of sensation in a limited area. Nerve impulse conduction is blocked by decreasing nerve cell membrane permeability to sodium ions, possibly by competing with calcium-binding sites that control sodium permeability. This change in permeability decreases depolarization and increases excitability threshold until the ability to generate an action potential is lost.
Pharmacology
Topical anesthetics are weak bases. They are made up of three important components: An aromatic ring, an intermediate length ester or amide linkage and a tertiary amine. The aromatic ring is primarily responsible for the lipid solubility that allows diffusion across the nerve cell membrane, determining the intrinsic property of these agents.[,] Protein binding of these agents depend on both the aromatic and amine portion.[]
Onset of action, anesthesia depth, and duration of action are determined by the pKa level, pH level, lipid solubility, protein binding, and vasodilatory effects of the specific local anesthetic. Other factors, which play important roles, are the site of application (faster onset at mucosa and sites with thin stratum corneum), vascularity of tissues in the area applied, the surface area, and duration of application.
Ester-type topical anesthetics are metabolized by plasma cholinesterase and other nonspecific esterases, while amide anesthetics are primarily metabolized in the liver via microsomal enzymes. Ester anesthetics are known to cause allergic manifestations on contact, while it is said to be a rare occurrence with amide anesthetics.[,] Para-amino benzoic acid (PABA), an ester hydrolysis metabolite is also known to be associated with allergic manifestations.[]
Skin Penetration Routes
There are three pathways to cross the stratum corneum, which is the main barrier for topical anesthetic agent delivery:[] Intercellular route (through the intercellular spaces of the cornified keratinocytes), para or transcellular route (through the cornified cells) and transappendageal route or shunt pathway (through the openings of the hair follicles and sweat glands) [Figure 1].
Skin penetration routes
Topical anesthetics are also able to penetrate mucosal surfaces, such as the mouth, genitals, and conjunctiva more easily than through a keratinized surface because of the absence of a stratum corneum.
Factors Determining the Dermal Drug DeliveryDrug form
Free bases are lipophilic and can penetrate the stratum corneum on its own whereas the salt forms require special delivery systems to do so.
Melting point and eutectic mixtures
The lower the melting point, the better the penetration is. Eutectic mixtures have a lower melting point, thus better penetration than either component by itself.
Concentration of drug in vehicle
Higher the concentration of drug in the vehicle, higher the rate of penetration.
Skin permeation enhancers
These compounds, promote skin permeability by increasing the permeability of the stratum corneum temporarily and reversibly. They can be:
Permeation enhancers under trial are eucalyptol, soya bean casein.[]
Physical means of enhancing permeation
Skin penetration of topically applied anesthetics can be enhanced by following physical measures:
Delivery from Lipid Vesicles
Liposomes, niosomes, and transfersomes are examples of lipid vesicles.
Liposomes are microscopic vesicles, which are composed of one or more lipid bilayers arranged in concentric fashion enclosing an equal number of aqueous compartments capable of entrapping lipid soluble or water-soluble drugs. The lipids used are typically phospholipids such as lecithin. Drug molecules can either be encapsulated in the aqueous space or intercalated into the lipid bilayer depending upon its physicochemical characteristics.[,] Studies with radioactive or fluorescence-labeled phospholipids have shown that the liposomes disperse in the upper layers of the stratum corneum, without further penetration into the epidermis, dermis or deeper.[] Fisher et al. in their study found that 5% liposome-encapsulated tetracaine produced better superficial local anesthesia than 5% eutectic mixture of local anesthetics (EMLA).[] Disadvantages of liposomes are their instability, and the predisposition of phospholipids to oxidative degradation.
Niosomes (microvesicles) are similar to liposomes, but are prepared from nonionic surfactants. They tend to be smaller in diameter than liposomes, and may have unilamellar (one layer), or multilamellar structures. They are more stable and may provide faster penetration to the stratum corneum than liposomes. They do not go deeper either.
Transfersomes are prepared using bile salt (sodium cholate) molecules. Unlike liposomes, transfersomes appear to be highly deformable and researchers claim that they can transport through pores, which are 5 times smaller than their size.
Various Topical PreparationsEutectic mixture of local anesthetics
Eutectic mixtures are compounds, which melt at lower temperatures than any of their components, permitting higher concentrations of anesthetics for use. It is 5% oil in water emulsion cream with a melting point of 18°C and consists of 25 mg/mL of lignocaine, 25 mg/mL of prilocaine, a thickener, an emulsifier, and distilled water adjusted to a pH level of 9.4. EMLA is applied in a thick layer (1-2 g/10 cm2, up to a maximal dose of 20 g/200 cm2) to intact skin. Pediatric dosing is shown in Table 1. After application, the area is covered with a patch of tegaderm or clear plastic wrap to facilitate penetration through the stratum corneum. Depth of anesthesia depends on the contact time with EMLA. Anesthetic effect has been shown to reach a maximal depth of 3 mm after a 60-min application, and 5 mm after a 120-min application. Dermal analgesia can be expected to increase for up to 3 h under occlusive dressing and persist for 1-2 h after removal of the cream. EMLA should not be applied to the palms and soles because of variable penetration. EMLA is a pregnancy category B agent, but caution should be exercised when being administered to a nursing mother, because lignocaine is excreted through breast milk (AstraZeneca insert).
Table 1Tetracaine, adrenaline (epinephrine), and cocaine (TAC)
Consists of 0.5% tetracaine, 0.05% adrenaline, and 11.8% cocaine. It was the first topical anesthetic mixture found to be effective for nonmucosal skin lacerations to the face and scalp. A dose of 1 ml/cm of laceration can be applied using a cotton-tipped applicator with firm pressure that is maintained for 20-40 min. However, it is no longer being used because of general concern about toxicity and expense, and federal regulatory issues involving medications containing cocaine.[]
Lidocaine, epinephrine, and tetracaine (LET)
Safer and more cost-effective alternative to TAC, contains 4% lignocaine with 0.1% epinephrine and 0.5% tetracaine. LET is used on nonmucosal skin lacerations by placing a few drops directly into the wound. A cotton-tipped applicator with 1-3 mL of the gel or solution is then applied directly to the wound with firm pressure for 15-30 min. It can be safely used in children older than 2 years of age. LET is slightly less effective on extremity lacerations. Because LET contains epinephrine, application to end-arteriolar parts of the body, such as the digits, should be avoided. Caution must also be exercised when contemplating the use of LET in contaminated wounds, complex wounds, or wounds larger than 6 cm. LET and TAC do not work on intact skin.[]
Bupivanor
It contains 0.48% bupivacaine and 1:26000 norepinephrine. Bupivanor is an effective alternative to TAC and lidocaine infiltration for local anesthesia during laceration repair, especially on the face and scalp.[]
ELA-max
It contains 4 or 5% (ELA-max 5) lignocaine cream in a liposomal matrix and is FDA-approved for the temporary relief of pain resulting from minor cuts and abrasions. ELA-max 5 is marketed for temporary relief of anorectal pain. ELA-max is applied to intact skin for 15-40 min with or without occlusion and provides a longer duration of anesthesia compared to nonliposomal preparations. Maximum area of application is 600 cm2. In children weighing less than 20 kg, a single application of ELA-max cream should not be applied to an area larger than 100 cm2.[]
Betacaine-LA
It contains lignocaine, prilocaine and phenylephrine. Betacaine-LA is a proprietary anesthetic and exact concentration of its ingredients is a trade secret. The pocket insert of the product reports concentrations of lignocaine and prilocaine to be 4 times that of EMLA and so, it should not be applied to an area larger than 300 cm2 in adults and is not advocated for use in children.[]
4% tetracaine (amethocaine)
It is a long acting ester anesthetic in lecithin gel base, with a recommended application time of 30-min under occlusive dressing and maximum dose limit of 50 mg.[]
Topicaine
Topicaine is 4% lignocaine in a gel microemulsion drug delivery system. The recommended application time by the manufacturers is 30-60 min. The maximum area of application is 600 cm2 in adults and 100 cm2 in children.[]
S-Caine Patch™ and local anesthetic peel
The patch (manufactured by ZARS, Inc., Salt Lake City, UT, US) contains a 1:1 eutectic mixture of 70 mg lignocaine and 70 mg tetracaine base, with a disposable, oxygen activated heating element, which helps in accelerating transcutaneous delivery and analgesic effect of local anesthetics. The heating element generates a controlled level of heating (39°C-41°C) over a period of 2 h.[,]
Lidoderm patch
Lidoderm is comprised of an adhesive material containing 5% lignocaine. Each adhesive patch contains 700 mg of lignocaine (50 mg/g adhesive) in an aqueous base. It has been recently approved by the FDA for the treatment of pain caused by postherpetic neuralgia.
Proparacaine or proxymetacaine
About 0.5% solution is suitable for ophthalmic use. With a single drop, the onset of anesthesia usually begins within 30 s, the maximum anesthetic effect is achieved at 5-min and duration of corneal anesthesia is 15-25 min.
Journal Of Anaesthesiology Clinical Pharmacology AbbreviationMiscellaneous agents with topical anesthetic potential
8-10% capsaicin[] (act on transient receptor potential vanilloid 1, i.e., the transient receptor potential channel of the vanilloid receptor family subtype 1); tetradotoxin,[,] 0.8% nalbuphine,[] ethyl chloride spray[] etc.
Clinical applications
Contraindications
Ester group topical anesthetics are contraindicated in patients with known allergy to PABA, sulfonamides and hair dyes.
Adverse effects
Burning or stinging at the administration site.
Systemic toxicity-due to excess dosage, repeated use, particularly in patients on risk like infants or children or elderly, or patients with liver disease etc. Manifestations can be as follows:
Because the risk of adverse events with improper application is real, physicians must exercise caution and good judgment while using topical anesthetics.
Conclusion
Topical anesthetics play an important role in decreasing the pain associated with ophthalmological, superficial dermatological, aesthetic and laser procedures, minor surgeries, venipuncture etc. With very wide varieties of agents and delivery devices being improvised upon every day, it seems time is not far off when we can completely abolish use of infiltrative local anesthesia. But, users should be well aware about the pharmacology of the agents being used and possible adverse events.
Footnotes
Source of Support: Nil
Conflicts of Interest: None declared.
European Journal Of AnesthesiologyReferences
1. Biscoping J, Bachmann-Mennenga MB. Local anesthetics from ester to isomer. Anasthesiol Intensivmed Notfallmed Schmerzther. 2000;35:285–92. [PubMed] [Google Scholar]
2. Jackson T, McLure HA. Pharmacology of local anesthetics. Ophthalmol Clin North Am. 2006;19:155–61. [PubMed] [Google Scholar]
3. Covino BG. Local anesthesia 1. N Engl J Med. 1972;286:975–83. [PubMed] [Google Scholar]
4. Covino BG. Local anesthesia 2. N Engl J Med. 1972;286:1035–42. [PubMed] [Google Scholar]
5. Covino BG. Local anesthetic agents for peripheral nerve blocks. Anaesthesist. 1980;29:33–7. [PubMed] [Google Scholar]
6. Covino BG. Physiology and pharmacology of local anesthetic agents. Anesth Prog. 1981;28:98–104.[PMC free article] [PubMed] [Google Scholar]
7. Suhonen R, Kanerva L. Contact allergy and cross-reactions caused by prilocaine. Am J Contact Dermat. 1997;8:231–5. [PubMed] [Google Scholar]
8. Fuzier R, Lapeyre-Mestre M, Mertes PM, Nicolas JF, Benoit Y, Didier A, et al. Immediate- and delayed-type allergic reactions to amide local anesthetics: Clinical features and skin testing. Pharmacoepidemiol Drug Saf. 2009;18:595–601. [PubMed] [Google Scholar]
9. Kluger N, Raison-Peyron N, Michot C, Guillot B, Bessis D. Acute bullous irritant contact dermatitis caused by EMLA ® cream. Contact Dermatitis. 2011;65:181–3. [PubMed] [Google Scholar]
10. Mackie BS, Mackie LE. The PABA story. Australas J Dermatol. 1999;40:51–3. [PubMed] [Google Scholar]
11. Adriani J, Dalili H. Penetration of local anesthetics through epithelial barriers. Anesth Analg. 1971;50:834–41. [PubMed] [Google Scholar]
12. Cázares-Delgadillo J, Naik A, Kalia YN, Quintanar-Guerrero D, Ganem-Quintanar A. Skin permeation enhancement by sucrose esters: A pH-dependent phenomenon. Int J Pharm. 2005;297:204–12. [PubMed] [Google Scholar]
13. Shokri J, Nokhodchi A, Dashbolaghi A, Hassan-Zadeh D, Ghafourian T, Barzegar Jalali M. The effect of surfactants on the skin penetration of diazepam. Int J Pharm. 2001;228:99–107. [PubMed] [Google Scholar]
14. Tian W, Hu Q, Xu Y, Xu Y. Effect of soybean-lecithin as an enhancer of buccal mucosa absorption of insulin. Biomed Mater Eng. 2012;22:171–8. [PubMed] [Google Scholar]
15. Shipton EA. New delivery systems for local anaesthetics-part 2. Anesthesiol Res Pract 2012. 2012 289373. [PMC free article] [PubMed] [Google Scholar]
16. Greenbaum SS. Iontophoresis as a tool for anesthesia in dermatologic surgery: An overview. Dermatol Surg. 2001;27:1027–30. [PubMed] [Google Scholar]
17. Kanebako M, Inagi T, Takayama K. Transdermal delivery of indomethacin by iontophoresis. Biol Pharm Bull. 2002;25:779–82. [PubMed] [Google Scholar]
18. Sammeta SM, Vaka SR, Murthy SN. Transdermal drug delivery enhanced by low voltage electropulsation (LVE) Pharm Dev Technol. 2009;14:159–64. [PubMed] [Google Scholar]
19. Wallace MS, Ridgeway B, Jun E, Schulteis G, Rabussay D, Zhang L. Topical delivery of lidocaine in healthy volunteers by electroporation, electroincorporation, or iontophoresis: An evaluation of skin anesthesia. Reg Anesth Pain Med. 2001;26:229–38. [PubMed] [Google Scholar]
20. Zempsky WT, Robbins B, McKay K. Reduction of topical anesthetic onset time using ultrasound: A randomized controlled trial prior to venipuncture in young children. Pain Med. 2008;9:795–802. [PubMed] [Google Scholar]
21. Sammeta SM, Repka MA, Narasimha Murthy S. Magnetophoresis in combination with chemical enhancers for transdermal drug delivery. Drug Dev Ind Pharm. 2011;37:1076–82.[PMC free article] [PubMed] [Google Scholar]
22. Baron ED, Harris L, Redpath WS, Shapiro H, Hetzel F, Morley G, et al. Laser-assisted penetration of topical anesthetic in adults. Arch Dermatol. 2003;139:1288–90. [PubMed] [Google Scholar]
23. Duan D, Moeckly C, Gysbers J, Novak C, Prochnow G, Siebenaler K, et al. Enhanced delivery of topically-applied formulations following skin pre-treatment with a hand-applied, plastic microneedle array. Curr Drug Deliv. 2011;8:557–65. [PubMed] [Google Scholar]
24. Gregoriadis G. The carrier potential of liposomes in biology and medicine (second of two parts) N Engl J Med. 1976;295:765–70. [PubMed] [Google Scholar]
25. Fendler JH, Romero A. Liposomes as drug carriers. Life Sci. 1977;20:1109–20. [PubMed] [Google Scholar]
26. Lieb LM, Ramachandran C, Egbaria K, Weiner N. Topical delivery enhancement with multilamellar liposomes into pilosebaceous units: I. In vitro evaluation using fluorescent techniques with the hamster ear model. J Invest Dermatol. 1992;99:108–13. [PubMed] [Google Scholar]
27. Fisher R, Hung O, Mezei M, Stewart R. Topical anaesthesia of intact skin: Liposome-encapsulated tetracaine vs EMLA. Br J Anaesth. 1998;81:972–3. [PubMed] [Google Scholar]
28. Kundu S, Achar S. Principles of office anesthesia: Part II. Topical anesthesia. Am Fam Physician. 2002;66:99–102. [PubMed] [Google Scholar]
29. Smith GA, Strausbaugh SD, Harbeck-Weber C, Shields BJ, Powers JD, Hackenberg D. Comparison of topical anesthetics without cocaine to tetracaine-adrenaline-cocaine and lidocaine infiltration during repair of lacerations: Bupivacaine-norepinephrine is an effective new topical anesthetic agent. Pediatrics. 1996;97:301–7. [PubMed] [Google Scholar]
30. Friedman PM, Mafong EA, Friedman ES, Geronemus RG. Topical anesthetics update: EMLA and beyond. Dermatol Surg. 2001;27:1019–26. [PubMed] [Google Scholar]
31. Sethna NF, Verghese ST, Hannallah RS, Solodiuk JC, Zurakowski D, Berde CB. A randomized controlled trial to evaluate S-Caine patch for reducing pain associated with vascular access in children. Anesthesiology. 2005;102:403–8. [PubMed] [Google Scholar]
32. Sawyer J, Febbraro S, Masud S, Ashburn MA, Campbell JC. Heated lidocaine/tetracaine patch (Synera, Rapydan) compared with lidocaine/prilocaine cream (EMLA) for topical anaesthesia before vascular access. Br J Anaesth. 2009;102:210–5. [PubMed] [Google Scholar]
33. Haanpää M, Treede RD. Capsaicin for neuropathic pain: Linking traditional medicine and molecular biology. Eur Neurol. 2012;68:264–75. [PubMed] [Google Scholar]
34. Schwartz DM, Fields HL, Duncan KG, Duncan JL, Jones MR. Experimental study of tetrodotoxin, a long-acting topical anesthetic. Am J Ophthalmol. 1998;125:481–7. [PubMed] [Google Scholar]
35. Wang L, Shankarappa SA, Tong R, Ciolino JB, Tsui JH, Chiang HH, et al. Topical drug formulations for prolonged corneal anesthesia. Cornea. 2013;32:1040–5.[PMC free article] [PubMed] [Google Scholar]
36. Lee CH, Lin SL, Chi TT, Chang SH, Wang HC. Effect of topical administration of 0.8% nalbuphine on the cornea in dogs after phacoemulsification. J Vet Med Sci. 2013;75:1041–7. [PubMed] [Google Scholar]
37. Soueid A, Richard B. Ethyl chloride as a cryoanalgesic in pediatrics for venipuncture. Pediatr Emerg Care. 2007;23:380–3. [PubMed] [Google Scholar]
38. Ruetzler K, Sima B, Mayer L, Golescu A, Dunkler D, Jaeger W, et al. Lidocaine/tetracaine patch (Rapydan) for topical anaesthesia before arterial access: A double-blind, randomized trial. Br J Anaesth. 2012;109:790–6. [PubMed] [Google Scholar]
39. Rauck R, Busch M, Marriott T. Effectiveness of a heated lidocaine/tetracaine topical patch for pain associated with myofascial trigger points: Results of an open-label pilot study. Pain Pract. 2013;13:533–8. [PubMed] [Google Scholar]
40. Vassilouthis J. Relief of trigeminal neuralgia by proparacaine. J Neurol Neurosurg Psychiatry. 1994;57:121.[PMC free article] [PubMed] [Google Scholar]
41. Spaziante R, Cappabianca P, Saini M, de Divitiis E. Topical ophthalmic treatment for trigeminal neuralgia. J Neurosurg. 1995;82:699–700. [PubMed] [Google Scholar]
42. Gambrell J, Schaal S. Topical anesthesia for intravitreal injection. Expert Opin Drug Deliv. 2012;9:731–3. [PubMed] [Google Scholar]
43. Jourdy DN, Kacker A. Regional anesthesia for office-based procedures in otorhinolaryngology. Anesthesiol Clin. 2010;28:457–68. [PubMed] [Google Scholar]
44. Weisskopf A. Phenol anesthesia for myringotomy. Laryngoscope. 1983;93:114. [PubMed] [Google Scholar]
45. Hoffman RA, Li CL. Tetracaine topical anesthesia for myringotomy. Laryngoscope. 2001;111:1636–8. [PubMed] [Google Scholar]
46. Luotonen J, Laitakari K, Karjalainen H, Jokinen K. EMLA in local anaesthesia of the tympanic membrane. Acta Otolaryngol Suppl. 1992;492:63–7. [PubMed] [Google Scholar]
47. Atighechi S, Baradaranfar MH, Akbari SA. Reduction of nasal bone fractures: A comparative study of general, local, and topical anesthesia techniques. J Craniofac Surg. 2009;20:382–4. [PubMed] [Google Scholar]
48. Resch K, Schilling C, Borchert BD, Klatzko M, Uden D. Topical anesthesia for pediatric lacerations: A randomized trial of lidocaine-epinephrine-tetracaine solution versus gel. Ann Emerg Med. 1998;32:693–7. [PubMed] [Google Scholar]
49. Hopkins CS, Buckley CJ, Bush GH. Pain-free injection in infants. Use of a lignocaine-prilocaine cream to prevent pain at intravenous induction of general anaesthesia in 1-5-year-old children. Anaesthesia. 1988;43:198–201. [PubMed] [Google Scholar]
50. Goodacre TE, Sanders R, Watts DA, Stoker M. Split skin grafting using topical local anaesthesia (EMLA): A comparison with infiltrated anaesthesia. Br J Plast Surg. 1988;41:533–8. [PubMed] [Google Scholar]
51. Gyftopoulos KI. The efficacy and safety of topical EMLA cream application for minor surgery of the adult penis. Urol Ann. 2012;4:145–9.[PMC free article] [PubMed] [Google Scholar]
52. Neal JM, Bernards CM, Butterworth JF, 4th, Di Gregorio G, Drasner K, Hejtmanek MR, et al. ASRA practice advisory on local anesthetic systemic toxicity. Reg Anesth Pain Med. 2010;35:152–61. [PubMed] [Google Scholar]
53. Adams V, Marley J, McCarroll C. Prilocaine induced methaemoglobinaemia in a medically compromised patient. Was this an inevitable consequence of the dose administered? Br Dent J. 2007;203:585–7. [PubMed] [Google Scholar]
54. Vallurupalli S, Manchanda S. Risk of acquired methemoglobinemia with different topical anesthetics during endoscopic procedures. Local Reg Anesth. 2011;4:25–8.[PMC free article] [PubMed] [Google Scholar]
55. Cortazzo JA, Lichtman AD. Methemoglobinemia: A Review and Recommendations for Management. J Cardiothorac Vasc Anesth. 2014;28:1055–9. [PubMed] [Google Scholar]
Articles from Journal of Anaesthesiology, Clinical Pharmacology are provided here courtesy of Wolters Kluwer -- Medknow Publications
Author information pack
Be advised that our average review period is now fewer than thirty days.
The Journal of Clinical Anesthesia (JCA) publishes definitive, peer-reviewed articles devoted to the clinical practice of anesthesia. The JCA should be a participant in continuing education of clinicians, forecast important issues and trends in anesthesia, and foster responsible debate on controversial issues. As an international journal, it encourages cross-communication between different cultures, informing our readers about medical and sometimes nonmedical journalism, and provides enjoyable reading. Advertising between the pages of editorial content is not permitted.
Submission checklist
You can use this list to carry out a final check of your submission before you send it to the journal for review. Please check the relevant section in this Guide for Authors for more details.
Ensure that the following items are present:
One author has been designated as the corresponding author with contact details:
• E-mail address • Full postal address
All necessary files have been uploaded:
Manuscript: • Include keywords • All figures (include relevant captions) • All tables (including titles, description, footnotes) • Ensure all figure and table citations in the text match the files provided • Indicate clearly if color should be used for any figures in print Graphical Abstracts / Highlights files (where applicable) Supplemental files (where applicable)
Further considerations
• Manuscript has been 'spell checked' and 'grammar checked' • All references mentioned in the Reference List are cited in the text, and vice versa • Permission has been obtained for use of copyrighted material from other sources (including the Internet) • A competing interests statement is provided, even if the authors have no competing interests to declare • Journal policies detailed in this guide have been reviewed • Referee suggestions and contact details provided, based on journal requirements
For further information, visit our Support Center.
Ethics in publishing
Please see our information pages on Ethics in publishing and Ethical guidelines for journal publication.
Declaration of interest
All authors must disclose any financial and personal relationships with other people or organizations that could inappropriately influence (bias) their work. Examples of potential competing interests include employment, consultancies, stock ownership, honoraria, paid expert testimony, patent applications/registrations, and grants or other funding. Authors must disclose any interests in two places: 1. A summary declaration of interest statement in the title page file (if double-blind) or the manuscript file (if single-blind). If there are no interests to declare then please state this: 'Declarations of interest: none'. This summary statement will be ultimately published if the article is accepted. 2. Detailed disclosures as part of a separate Declaration of Interest form, which forms part of the journal's official records. It is important for potential interests to be declared in both places and that the information matches. More information. Funding sources and corporate or institutional associations must be acknowledged on the title page of the submitted article. In the cover letter accompanying the manuscript, provide the names of associations, consultancies, individual philanthropists, stock ownership, or other equity interest (e.g., patent or licensing arrangements). If the manuscript is accepted for publication, disclosure of this information will be discussed with the author(s).
Submission declaration and verification
Submission of an article implies that the work described has not been published previously (except in the form of an abstract, a published lecture or academic thesis, see 'Multiple, redundant or concurrent publication' for more information), that it is not under consideration for publication elsewhere, that its publication is approved by all authors and tacitly or explicitly by the responsible authorities where the work was carried out, and that, if accepted, it will not be published elsewhere in the same form, in English or in any other language, including electronically without the written consent of the copyright-holder. To verify originality, your article may be checked by the originality detection service Crossref Similarity Check.
Preprints
Please note that preprints can be shared anywhere at any time, in line with Elsevier's sharing policy. Sharing your preprints e.g. on a preprint server will not count as prior publication (see 'Multiple, redundant or concurrent publication' for more information).
Use of inclusive language
Inclusive language acknowledges diversity, conveys respect to all people, is sensitive to differences, and promotes equal opportunities. Articles should make no assumptions about the beliefs or commitments of any reader, should contain nothing which might imply that one individual is superior to another on the grounds of race, sex, culture or any other characteristic, and should use inclusive language throughout. Authors should ensure that writing is free from bias, for instance by using 'he or she', 'his/her' instead of 'he' or 'his', and by making use of job titles that are free of stereotyping (e.g. 'chairperson' instead of 'chairman' and 'flight attendant' instead of 'stewardess').
Authorship
All authors should have made substantial contributions to all of the following: (1) the conception and design of the study, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, (3) final approval of the version to be submitted.
Changes to authorship
Authors are expected to consider carefully the list and order of authors before submitting their manuscript and provide the definitive list of authors at the time of the original submission. Any addition, deletion or rearrangement of author names in the authorship list should be made only before the manuscript has been accepted and only if approved by the journal Editor. To request such a change, the Editor must receive the following from the corresponding author: (a) the reason for the change in author list and (b) written confirmation (e-mail, letter) from all authors that they agree with the addition, removal or rearrangement. In the case of addition or removal of authors, this includes confirmation from the author being added or removed. Only in exceptional circumstances will the Editor consider the addition, deletion or rearrangement of authors after the manuscript has been accepted. While the Editor considers the request, publication of the manuscript will be suspended. If the manuscript has already been published in an online issue, any requests approved by the Editor will result in a corrigendum.
Copyright
Upon acceptance of an article, authors will be asked to complete a 'Journal Publishing Agreement' (see more information on this). An e-mail will be sent to the corresponding author confirming receipt of the manuscript together with a 'Journal Publishing Agreement' form or a link to the online version of this agreement.
Subscribers may reproduce tables of contents or prepare lists of articles including abstracts for internal circulation within their institutions. Permission of the Publisher is required for resale or distribution outside the institution and for all other derivative works, including compilations and translations. If excerpts from other copyrighted works are included, the author(s) must obtain written permission from the copyright owners and credit the source(s) in the article. Elsevier has preprinted forms for use by authors in these cases.
For gold open access articles: Upon acceptance of an article, authors will be asked to complete an 'Exclusive License Agreement' (more information). Permitted third party reuse of gold open access articles is determined by the author's choice of user license.
Author rights
As an author you (or your employer or institution) have certain rights to reuse your work. More information.
Acceptable Secondary Publication
Secondary publication in the same or another language, especially in other countries, is justifiable, and can be beneficial, provided all of the following conditions are met:
Permission for such secondary publication should be free of charge.
Elsevier supports responsible sharing
Find out how you can share your research published in Elsevier journals.
Role of the funding source
You are requested to identify who provided financial support for the conduct of the research and/or preparation of the article and to briefly describe the role of the sponsor(s), if any, in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication. If the funding source(s) had no such involvement then this should be stated.
Funding body agreements and policies
Elsevier has established a number of agreements with funding bodies which allow authors to comply with their funder's open access policies. Some funding bodies will reimburse the author for the gold open access publication fee. Details of existing agreements are available online. After acceptance, open access papers will be published under a noncommercial license. For authors requiring a commercial CC BY license, you can apply after your manuscript is accepted for publication.
Open access
This journal offers authors a choice in publishing their research:
Subscription
• Articles are made available to subscribers as well as developing countries and patient groups through our universal access programs. • No open access publication fee payable by authors. • The Author is entitled to post the accepted manuscript in their institution's repository and make this public after an embargo period (known as green Open Access). The published journal article cannot be shared publicly, for example on ResearchGate or Academia.edu, to ensure the sustainability of peer-reviewed research in journal publications. The embargo period for this journal can be found below. Gold open access • Articles are freely available to both subscribers and the wider public with permitted reuse. • A gold open access publication fee is payable by authors or on their behalf, e.g. by their research funder or institution.
Regardless of how you choose to publish your article, the journal will apply the same peer review criteria and acceptance standards.
For gold open access articles, permitted third party (re)use is defined by the following Creative Commons user licenses:
Creative Commons Attribution-NonCommercial-NoDerivs (CC BY-NC-ND)
For non-commercial purposes, lets others distribute and copy the article, and to include in a collective work (such as an anthology), as long as they credit the author(s) and provided they do not alter or modify the article. The gold open access publication fee for this journal is USD 2500, excluding taxes. Learn more about Elsevier's pricing policy: https://www.elsevier.com/openaccesspricing.
Green open access
Authors can share their research in a variety of different ways and Elsevier has a number of green open access options available. We recommend authors see our open access page for further information. Authors can also self-archive their manuscripts immediately and enable public access from their institution's repository after an embargo period. This is the version that has been accepted for publication and which typically includes author-incorporated changes suggested during submission, peer review and in editor-author communications. Embargo period: For subscription articles, an appropriate amount of time is needed for journals to deliver value to subscribing customers before an article becomes freely available to the public. This is the embargo period and it begins from the date the article is formally published online in its final and fully citable form. Find out more. This journal has an embargo period of 12 months.
Elsevier Researcher Academy
Researcher Academy is a free e-learning platform designed to support early and mid-career researchers throughout their research journey. The 'Learn' environment at Researcher Academy offers several interactive modules, webinars, downloadable guides and resources to guide you through the process of writing for research and going through peer review. Feel free to use these free resources to improve your submission and navigate the publication process with ease.
Language (usage and editing services)
Please write your text in good English (American or British usage is accepted, but not a mixture of these). Authors who feel their English language manuscript may require editing to eliminate possible grammatical or spelling errors and to conform to correct scientific English may wish to use the English Language Editing service available from Elsevier's WebShop.
Informed consent and patient details
Studies on patients or volunteers require ethics committee approval and informed consent, which should be documented in the paper. Appropriate consents, permissions and releases must be obtained where an author wishes to include case details or other personal information or images of patients and any other individuals in an Elsevier publication. Written consents must be retained by the author but copies should not be provided to the journal. Only if specifically requested by the journal in exceptional circumstances (for example if a legal issue arises) the author must provide copies of the consents or evidence that such consents have been obtained. For more information, please review the Elsevier Policy on the Use of Images or Personal Information of Patients or other Individuals. Unless you have written permission from the patient (or, where applicable, the next of kin), the personal details of any patient included in any part of the article and in any supplementary materials (including all illustrations and videos) must be removed before submission. In all manuscripts reporting the results of human studies, a statement must appear in the Materials and Methods section indicating that approval was obtained from the institutional review board (IRB) and that all human subjects signed written informed consent. Cite in the text the full (hospital) name of the IRB.
Submission
Our online submission system guides you stepwise through the process of entering your article details and uploading your files. The system converts your article files to a single PDF file used in the peer-review process. Editable files (e.g., Word, LaTeX) are required to typeset your article for final publication. All correspondence, including notification of the Editor's decision and requests for revision, is sent by e-mail.
Submit your article
Please submit your article via http://www.evise.com/evise/jrnl/JCA. Authors who are unable to provide an electronic version or have other circumstances that prevent online submission must contact the Editorial Office prior to submission to discuss alternate options. The Publisher and Editors regret that they are not able to consider submissions that do not follow these procedures.
Manuscript submission preparation
Authors must upload the following items as separate files: a cover letter, an abstract, the manuscript, and any tables, figures, and figure legends. Revised or resubmitted manuscripts should also include a transmittal letter, explaining how the author has dealt with each of the reviewer's and Editor's comments, and this letter should be uploaded as a 'Revision Comments' file on the system. • Provide a cover letter indicating the name, address, tel., fax, and e-mail addresses of the individual to whom correspondence and questions should be directed. Include a statement that the contents have not been published elsewhere and the paper is not being submitted elsewhere. Also indicate that the manuscript has been read and approved by all co-authors. Finally, indicate to which category of publication the author is submitting the article: Original Contribution/Research Paper, Editorial, Case Report, Pharmacologic Review, Special Article (history, art, politics, etc.), or Letter to the Editor. • Arrange the manuscript in the following order: title page, abstract (structured abstract if the work is an Original contribution only), text, acknowledgments, references, tables, illustrations, and legends for illustrations. • Text: Arrange the body of the manuscript in the following order, with each component beginning on a new page: Abstract, Introduction, Materials and Methods, Results, Discussion. • Number the pages in sequence, with the title page as page 1, the (structured) abstract as page 2, etc., with the page number in the upper right-hand corner.
Peer review
This journal operates a single blind review process. All contributions will be initially assessed by the editor for suitability for the journal. Papers deemed suitable are then typically sent to a minimum of two independent expert reviewers to assess the scientific quality of the paper. The Editor is responsible for the final decision regarding acceptance or rejection of articles. The Editor's decision is final. More information on types of peer review.
Use of word processing software
It is important that the file be saved in the native format of the word processor used. The text should be in single-column format. Keep the layout of the text as simple as possible. Most formatting codes will be removed and replaced on processing the article. In particular, do not use the word processor's options to justify text or to hyphenate words. However, do use bold face, italics, subscripts, superscripts etc. When preparing tables, if you are using a table grid, use only one grid for each individual table and not a grid for each row. If no grid is used, use tabs, not spaces, to align columns. The electronic text should be prepared in a way very similar to that of conventional manuscripts (see also the Guide to Publishing with Elsevier). Note that source files of figures, tables and text graphics will be required whether or not you embed your figures in the text. See also the section on Electronic artwork. To avoid unnecessary errors you are strongly advised to use the 'spell-check' and 'grammar-check' functions of your word processor.
Article structure
Subdivision - numbered sections
Divide your article into clearly defined and numbered sections. Subsections should be numbered 1.1 (then 1.1.1, 1.1.2, ..), 1.2, etc. (the abstract is not included in section numbering). Use this numbering also for internal cross-referencing: do not just refer to 'the text'. Any subsection may be given a brief heading. Each heading should appear on its own separate line.
Introduction
State the objectives of the work and provide an adequate background, avoiding a detailed literature survey or a summary of the results.
Materials and Methods
A clear and precise description of the experiment and the subjects from whom the data were derived. If subjects were randomized, describe the methods of randomization used. Identify all drugs and chemicals used, dosages, and routes of administration. Define all statistical methods used and, if appropriate, statistical references. Identify the name and address of the statistician who reviewed the data, if appropriate.
Results
A factual account of the study's findings. Present these as logically appropriate in text, tables, or illustrations; do not repeat in the text what is demonstrated in a table or illustration.
Discussion
A summary emphasizing new and important aspects of the study's conclusions. Ensure that all conclusions are justified by the results of the study. Identify the implications of the findings and their limitations, including implications for future research. Look at the pros and cons of the methods and results. Compare the implications and limitations of these findings with those reported in other essential studies. State new hypotheses when warranted, but clearly label them as such. Recommendations, where appropriate, may be included.
Appendices
If there is more than one appendix, they should be identified as A, B, etc. Formulae and equations in appendices should be given separate numbering: Eq. (A.1), Eq. (A.2), etc.; in a subsequent appendix, Eq. (B.1) and so on. Similarly for tables and figures: Table A.1; Fig. A.1, etc.
Essential Title Page Information
• Title. Concise and informative. Titles are often used in information-retrieval systems. Avoid abbreviations and formulae where possible. • Author names and affiliations. Give the first name, middle initial, and last name of all authors. Where the family name may be ambiguous (e.g., a double name), please indicate this clearly. List each author's highest academic degree in parentheses after their name. Present the authors' affiliation addresses (where the actual work was done) below the names. Indicate all affiliations with a lower-case superscript letter immediately after the author's name and in front of the appropriate address. Provide the full postal address of each affiliation, including the country name and, if available, the e-mail address of each author. • Corresponding author. Clearly indicate who will handle correspondence at all stages of refereeing and publication, also post-publication. Ensure that phone numbers (with country and area code) are provided in addition to the e-mail address and the complete postal address. Contact details must be kept up to date by the corresponding author. • Present/permanent address. If an author has moved since the work described in the article was done, or was visiting at the time, a 'Present address' (or 'Permanent address') may be indicated as a footnote to that author's name. The address at which the author actually did the work must be retained as the main, affiliation address. Superscript Arabic numerals are used for such footnotes. •Running title. Provide a running title of up to 50 characters. •Disclosures. Acknowledge grants, sponsors, and funding sources that provided direct financial support to the research work contained in the manuscript.
Highlights
Highlights are mandatory for this journal. They consist of a short collection of bullet points that convey the core findings of the article and should be submitted in a separate editable file in the online submission system. Please use 'Highlights' in the file name and include 3 to 5 bullet points (maximum 85 characters, including spaces, per bullet point). You can view example Highlights on our information site.
Abstract
A concise and factual abstract is required for all article types except Editorials and Letters to the Editor. The abstract should state briefly the purpose of the research, the principal results and major conclusions. An abstract is often presented separately from the article, so it must be able to stand alone. For this reason, References should be avoided, but if essential, then cite the author(s) and year(s). Also, non-standard or uncommon abbreviations should be avoided, but if essential they must be defined at their first mention in the abstract itself. All Original Contributions require a structured abstract of up to 300 words, which clearly states the following (use these subheadings): • Study Objective. • Design. The type of study conducted. • Setting. e.g., delivery room, postoperative recovery area, operating room • Patients. i.e., number studied, their ASA physical status, the nature of their procedure and illness • Interventions. What was done to the patients on behalf of the study. • Measurements e.g., blood pressure, heart rate, resp. rate values collected • Main Results. The study's principal finding of interest. • Conclusions. Based on the kinds of patients studied and the interventions used, what conclusions may be drawn from the main findings of interest. The structured abstract should provide key information and be informative on its own, although it should not include discussion of pros and cons, limitations, or justifications of the study.
Graphical abstract
Although a graphical abstract is optional, its use is encouraged as it draws more attention to the online article. The graphical abstract should summarize the contents of the article in a concise, pictorial form designed to capture the attention of a wide readership. Graphical abstracts should be submitted as a separate file in the online submission system. Image size: Please provide an image with a minimum of 531 × 1328 pixels (h × w) or proportionally more. The image should be readable at a size of 5 × 13 cm using a regular screen resolution of 96 dpi. Preferred file types: TIFF, EPS, PDF or MS Office files. You can view Example Graphical Abstracts on our information site. Authors can make use of Elsevier's Illustration Services to ensure the best presentation of their images and in accordance with all technical requirements.
Keywords
Immediately after the abstract, provide a maximum of 6 keywords, using American spelling and avoiding general and plural terms and multiple concepts (avoid, for example, 'and', 'of'). Be sparing with abbreviations: only abbreviations firmly established in the field may be eligible.Use terms from the U.S. Library of Medicine's Medical Subject Headings (MeSH) Annotated Alphabetical List for indexing purposes in Index Medicus.
Acknowledgements
Collate acknowledgements in a separate section at the end of the article before the references and do not, therefore, include them on the title page, as a footnote to the title or otherwise. List here those individuals who provided help during the research (e.g., assisted with the study or with manuscript preparation, including statistical review, by providing language help, writing assistance, or proof reading the article, etc.).
Formatting of funding sources
List funding sources in this standard way to facilitate compliance to funder's requirements:
Funding: This work was supported by the National Institutes of Health [grant numbers xxxx, yyyy]; the Bill & Melinda Gates Foundation, Seattle, WA [grant number zzzz]; and the United States Institutes of Peace [grant number aaaa].
It is not necessary to include detailed descriptions on the program or type of grants and awards. When funding is from a block grant or other resources available to a university, college, or other research institution, submit the name of the institute or organization that provided the funding.
If no funding has been provided for the research, please include the following sentence:
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Units
Follow internationally accepted rules and conventions: use the international system of units (SI). If other units are mentioned, please give their equivalent in SI.
Math formulae
Please submit math equations as editable text and not as images. Present simple formulae in line with normal text where possible and use the solidus (/) instead of a horizontal line for small fractional terms, e.g., X/Y. In principle, variables are to be presented in italics. Powers of e are often more conveniently denoted by exp. Number consecutively any equations that have to be displayed separately from the text (if referred to explicitly in the text).
Footnotes
Footnotes should be used sparingly. Number them consecutively throughout the article. Many word processors can build footnotes into the text, and this feature may be used. Otherwise, please indicate the position of footnotes in the text and list the footnotes themselves separately at the end of the article. Do not include footnotes in the Reference list.
Artwork
Electronic artwork
General points • Make sure you use uniform lettering and sizing of your original artwork. • Embed the used fonts if the application provides that option. • Aim to use the following fonts in your illustrations: Arial, Courier, Times New Roman, Symbol, or use fonts that look similar. • Number the illustrations according to their sequence in the text. • Use a logical naming convention for your artwork files. • Provide captions to illustrations separately. • Size the illustrations close to the desired dimensions of the published version. • Submit each illustration as a separate file. A detailed guide on electronic artwork is available. You are urged to visit this site; some excerpts from the detailed information are given here. Formats If your electronic artwork is created in a Microsoft Office application (Word, PowerPoint, Excel) then please supply 'as is' in the native document format. Regardless of the application used other than Microsoft Office, when your electronic artwork is finalized, please 'Save as' or convert the images to one of the following formats (note the resolution requirements for line drawings, halftones, and line/halftone combinations given below): EPS (or PDF): Vector drawings, embed all used fonts. TIFF (or JPEG): Color or grayscale photographs (halftones), keep to a minimum of 300 dpi. TIFF (or JPEG): Bitmapped (pure black & white pixels) line drawings, keep to a minimum of 1000 dpi. TIFF (or JPEG): Combinations bitmapped line/half-tone (color or grayscale), keep to a minimum of 500 dpi. Please do not: • Supply files that are optimized for screen use (e.g., GIF, BMP, PICT, WPG); these typically have a low number of pixels and limited set of colors; • Supply files that are too low in resolution; • Submit graphics that are disproportionately large for the content.
Further considerations
• A figure and its legend should be sufficiently informative that the results can be understood without reference to the text. In the legend, identify and define all abbreviations, symbols, arrows, and illustration parts. • The preferred point symbols are open circle, open square, open triangle, filled circle, filled square, filled triangle. The preferred shadings are black, white and bold hatching. Avoid stippling, which does not reproduce well. The preferred font for axis labels and keys is Arial. • Ticks should be drawn outside the figure axes; they should not be extended to form lines across the whole figure. • Number figures consecutively in Arabic numerals. • In the editorial text, abbreviate 'Figure' to 'Fig.' and 'Figures' to 'Figs.' except when starting a sentence.
Color artwork
Please make sure that artwork files are in an acceptable format (TIFF (or JPEG), EPS (or PDF), or MS Office files) and with the correct resolution. If, together with your accepted article, you submit usable color figures then Elsevier will ensure, at no additional charge, that these figures will appear in color online (e.g., ScienceDirect and other sites) regardless of whether or not these illustrations are reproduced in color in the printed version. For color reproduction in print, you will receive information regarding the costs from Elsevier after receipt of your accepted article. Please indicate your preference for color: in print or online only. Further information on the preparation of electronic artwork.
Illustration services
Elsevier's WebShop offers Illustration Services to authors preparing to submit a manuscript but concerned about the quality of the images accompanying their article. Elsevier's expert illustrators can produce scientific, technical and medical-style images, as well as a full range of charts, tables and graphs. Image 'polishing' is also available, where our illustrators take your image(s) and improve them to a professional standard. Please visit the website to find out more.
Tables
Each table should be uploaded as a separate file. Number tables consecutively in accordance with their appearance in the text. Abbreviations may be used to simplify a table; define all abbreviations in a single footnote. Place footnotes to tables below the table body and indicate them with superscript lowercase letters. Identify statistical measures of variation such as standard deviation and standard error of the mean. Avoid vertical rules. Be sparing in the use of tables and ensure that the data presented in tables do not duplicate results described elsewhere in the article.
References
Citation in text
Please ensure that every reference cited in the text is also present in the reference list (and vice versa). Any references cited in the abstract must be given in full. Unpublished results and personal communications are not recommended in the reference list, but may be mentioned in the text. If these references are included in the reference list they should follow the standard reference style of the journal and should include a substitution of the publication date with either 'Unpublished results' or 'Personal communication'. Citation of a reference as 'in press' implies that the item has been accepted for publication.
Reference links
Increased discoverability of research and high quality peer review are ensured by online links to the sources cited. In order to allow us to create links to abstracting and indexing services, such as Scopus, CrossRef and PubMed, please ensure that data provided in the references are correct. Please note that incorrect surnames, journal/book titles, publication year and pagination may prevent link creation. When copying references, please be careful as they may already contain errors. Use of the DOI is highly encouraged.
A DOI is guaranteed never to change, so you can use it as a permanent link to any electronic article. An example of a citation using DOI for an article not yet in an issue is: VanDecar J.C., Russo R.M., James D.E., Ambeh W.B., Franke M. (2003). Aseismic continuation of the Lesser Antilles slab beneath northeastern Venezuela. Journal of Geophysical Research, https://doi.org/10.1029/2001JB000884. Please note the format of such citations should be in the same style as all other references in the paper.
Web references
As a minimum, the full URL should be given and the date when the reference was last accessed. Any further information, if known (DOI, author names, dates, reference to a source publication, etc.), should also be given. Web references can be listed separately (e.g., after the reference list) under a different heading if desired, or can be included in the reference list.
Data references
This journal encourages you to cite underlying or relevant datasets in your manuscript by citing them in your text and including a data reference in your Reference List. Data references should include the following elements: author name(s), dataset title, data repository, version (where available), year, and global persistent identifier. Add [dataset] immediately before the reference so we can properly identify it as a data reference. The [dataset] identifier will not appear in your published article.
Reference management software
Most Elsevier journals have their reference template available in many of the most popular reference management software products. These include all products that support Citation Style Language styles, such as Mendeley. Using citation plug-ins from these products, authors only need to select the appropriate journal template when preparing their article, after which citations and bibliographies will be automatically formatted in the journal's style. If no template is yet available for this journal, please follow the format of the sample references and citations as shown in this Guide. If you use reference management software, please ensure that you remove all field codes before submitting the electronic manuscript. More information on how to remove field codes from different reference management software. Users of Mendeley Desktop can easily install the reference style for this journal by clicking the following link: http://open.mendeley.com/use-citation-style/journal-of-clinical-anesthesia When preparing your manuscript, you will then be able to select this style using the Mendeley plug-ins for Microsoft Word or LibreOffice.
Reference style
Text: Indicate references by number(s) in square brackets in line with the text. The actual authors can be referred to, but the reference number(s) must always be given. List: Number the references (numbers in square brackets) in the list in the order in which they appear in the text. Examples: Reference to a journal publication: [1] Van der Geer J, Hanraads JAJ, Lupton RA. The art of writing a scientific article. J Sci Commun 2010;163:51–9. https://doi.org/10.1016/j.Sc.2010.00372. Reference to a journal publication with an article number: [2] Van der Geer J, Hanraads JAJ, Lupton RA. The art of writing a scientific article. Heliyon. 2018;19:e00205. https://doi.org/10.1016/j.heliyon.2018.e00205 Reference to a book: [3] Strunk Jr W, White EB. The elements of style. 4th ed. New York: Longman; 2000. Reference to a chapter in an edited book: [4] Mettam GR, Adams LB. How to prepare an electronic version of your article. In: Jones BS, Smith RZ, editors. Introduction to the electronic age, New York: E-Publishing Inc; 2009, p. 281–304. Reference to a website: [5] Cancer Research UK. Cancer statistics reports for the UK, http://www.cancerresearchuk.org/aboutcancer/statistics/cancerstatsreport/; 2003 [accessed 13 March 2003]. Reference to a dataset: [dataset] [6] Oguro M, Imahiro S, Saito S, Nakashizuka T. Mortality data for Japanese oak wilt disease and surrounding forest compositions, Mendeley Data, v1; 2015. https://doi.org/10.17632/xwj98nb39r.1. Note shortened form for last page number. e.g., 51–9, and that for more than 6 authors the first 6 should be listed followed by 'et al.' For further details you are referred to 'Uniform Requirements for Manuscripts submitted to Biomedical Journals' (J Am Med Assoc 1997;277:927–34) (see also Samples of Formatted References).
Video
Elsevier accepts video material and animation sequences to support and enhance your scientific research. Authors who have video or animation files that they wish to submit with their article are strongly encouraged to include links to these within the body of the article. This can be done in the same way as a figure or table by referring to the video or animation content and noting in the body text where it should be placed. All submitted files should be properly labeled so that they directly relate to the video file's content. . In order to ensure that your video or animation material is directly usable, please provide the file in one of our recommended file formats with a preferred maximum size of 150 MB per file, 1 GB in total. Video and animation files supplied will be published online in the electronic version of your article in Elsevier Web products, including ScienceDirect. Please supply 'stills' with your files: you can choose any frame from the video or animation or make a separate image. These will be used instead of standard icons and will personalize the link to your video data. For more detailed instructions please visit our video instruction pages. Note: since video and animation cannot be embedded in the print version of the journal, please provide text for both the electronic and the print version for the portions of the article that refer to this content.
Data visualization
Include interactive data visualizations in your publication and let your readers interact and engage more closely with your research. Follow the instructions here to find out about available data visualization options and how to include them with your article.
Supplementary material
Supplementary material such as applications, images and sound clips, can be published with your article to enhance it. Submitted supplementary items are published exactly as they are received (Excel or PowerPoint files will appear as such online). Please submit your material together with the article and supply a concise, descriptive caption for each supplementary file. If you wish to make changes to supplementary material during any stage of the process, please make sure to provide an updated file. Do not annotate any corrections on a previous version. Please switch off the 'Track Changes' option in Microsoft Office files as these will appear in the published version.
Research data
This journal encourages and enables you to share data that supports your research publication where appropriate, and enables you to interlink the data with your published articles. Research data refers to the results of observations or experimentation that validate research findings. To facilitate reproducibility and data reuse, this journal also encourages you to share your software, code, models, algorithms, protocols, methods and other useful materials related to the project.
Below are a number of ways in which you can associate data with your article or make a statement about the availability of your data when submitting your manuscript. If you are sharing data in one of these ways, you are encouraged to cite the data in your manuscript and reference list. Please refer to the 'References' section for more information about data citation. For more information on depositing, sharing and using research data and other relevant research materials, visit the research data page.
Data linking
If you have made your research data available in a data repository, you can link your article directly to the dataset. Elsevier collaborates with a number of repositories to link articles on ScienceDirect with relevant repositories, giving readers access to underlying data that gives them a better understanding of the research described.
There are different ways to link your datasets to your article. When available, you can directly link your dataset to your article by providing the relevant information in the submission system. For more information, visit the database linking page.
For supported data repositories a repository banner will automatically appear next to your published article on ScienceDirect.
In addition, you can link to relevant data or entities through identifiers within the text of your manuscript, using the following format: Database: xxxx (e.g., TAIR: AT1G01020; CCDC: 734053; PDB: 1XFN).
Mendeley Data
This journal supports Mendeley Data, enabling you to deposit any research data (including raw and processed data, video, code, software, algorithms, protocols, and methods) associated with your manuscript in a free-to-use, open access repository. During the submission process, after uploading your manuscript, you will have the opportunity to upload your relevant datasets directly to Mendeley Data. The datasets will be listed and directly accessible to readers next to your published article online.
For more information, visit the Mendeley Data for journals page.
Data statement
To foster transparency, we encourage you to state the availability of your data in your submission. This may be a requirement of your funding body or institution. If your data is unavailable to access or unsuitable to post, you will have the opportunity to indicate why during the submission process, for example by stating that the research data is confidential. The statement will appear with your published article on ScienceDirect. For more information, visit the Data Statement page.
Formatting of text
• Type all manuscripts with double line spacing and aligned left, including the abstract, references, and figure legends. • Use Times New Roman 12-point font/size. • Manuscripts should have continuous line numbers, page numbers, and wide margins throughout (including the abstract, references, figure legends, and tables). • Indent each new paragraph. • Use two returns to end headings and paragraphs. • Do not use lower-case 'l' (el) for '1' (one) or 'O'(oh) for '0' (zero); they have different typesetting values.
Online proof correction
Corresponding authors will receive an e-mail with a link to our online proofing system, allowing annotation and correction of proofs online. The environment is similar to MS Word: in addition to editing text, you can also comment on figures/tables and answer questions from the Copy Editor. Web-based proofing provides a faster and less error-prone process by allowing you to directly type your corrections, eliminating the potential introduction of errors. If preferred, you can still choose to annotate and upload your edits on the PDF version. All instructions for proofing will be given in the e-mail we send to authors, including alternative methods to the online version and PDF. We will do everything possible to get your article published quickly and accurately. Please use this proof only for checking the typesetting, editing, completeness and correctness of the text, tables and figures. Significant changes to the article as accepted for publication will only be considered at this stage with permission from the Editor. It is important to ensure that all corrections are sent back to us in one communication. Please check carefully before replying, as inclusion of any subsequent corrections cannot be guaranteed. Proofreading is solely your responsibility.
Additional Information
The Journal's Editor-in-Chief is Robert R. Gaiser, MD. Direct questions to the editorial office at (508) 540-2197; Fax (508) 540-2714; E-mail [email protected]. Visit the Elsevier Support Center to find the answers you need. Here you will find everything from Frequently Asked Questions to ways to get in touch. You can also check the status of your submitted article or find out when your accepted article will be published.
(Redirected from Journal of Anaesthesiology Clinical Pharmacology)
This is a list of academic journals published by Medknow Publications.
A[edit]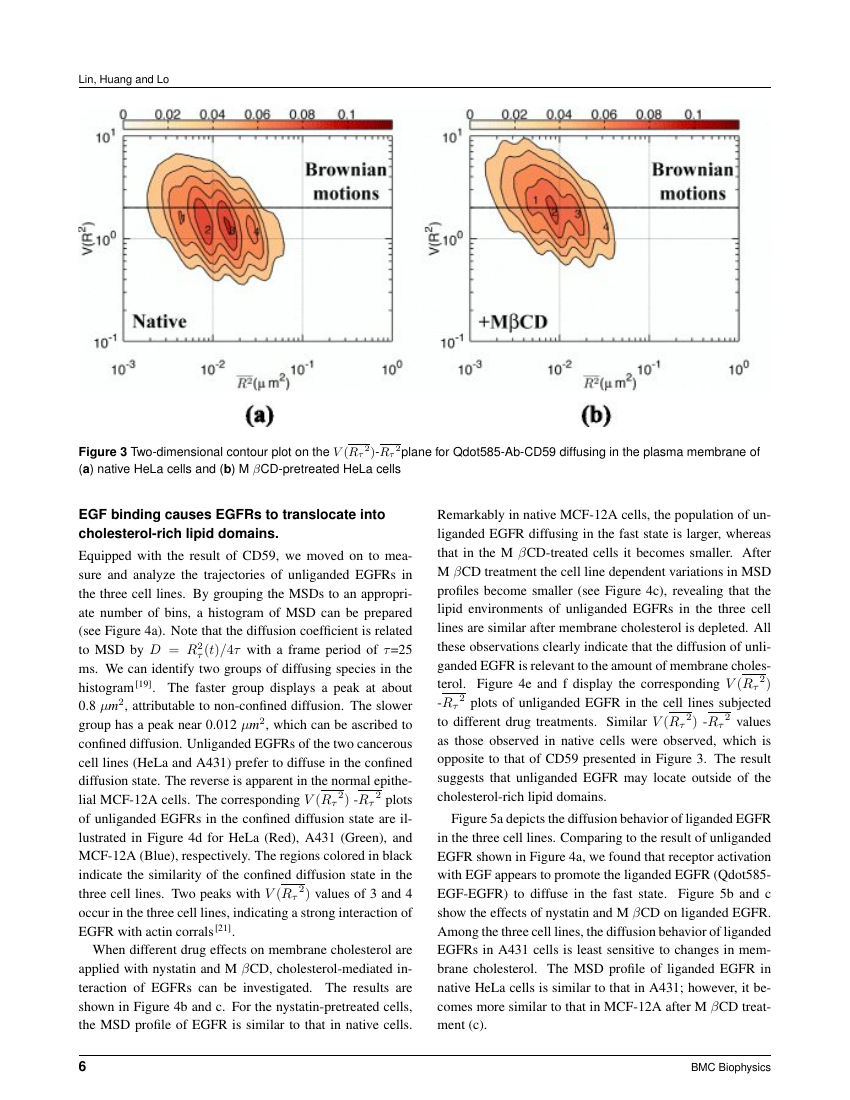
B[edit]
C[edit]
D[edit]
E[edit]
F[edit]
G[edit]
H[edit]
I[edit]
J[edit]
K[edit]
L[edit]
M[edit]
N[edit]
O[edit]
P[edit]
Q[edit]
R[edit]
S[edit]
T[edit]
U[edit]
V[edit]W[edit]
Y[edit]External links[edit]
Retrieved from 'https://en.wikipedia.org/w/index.php?title=List_of_Medknow_Publications_academic_journals&oldid=889286281'
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Home
- New Page
- New Page
- About Me
- Blog
- Il2 Cliffs Of Dover
- Fallout 4 Handcuff Mod
- Bmw Video In Motiuon
- Silent Legion Armor Conan Exiles
- Metal Gear Solid 3 Guide Pdf
- Download Driver Usb Gps Garmin 76csx
- Far Cry 3 Download
- Change Pdf To Jpg
- Oblivion Better Looking Armor
- Half Life 2 Ep 2 Soundtrack
- Serial Number Corel X7
- Car Speaker And Amplifier Matching Guide
- Baixar Discografia Kenny Roger
- Only Easy Day Was Yesterday
- Contact
- How To Disable Incognito On Chrome
- How To Add Razer Kraken To Razer Synapse
- How To Download Sims In Sims 4
- Shl Opq Practice Test
- Excel Button To Change Data On Dashbord
- Lake County Fl Jail Mugshots
- Star Wars Republic Commando Delta Squad
- Real Steel Games Free Download
 RSS Feed
RSS Feed
